Encorafenib in combination with Cetuximab in BRAFV600E–Mutated Metastatic Colorectal Cancer: A European pooled analysis of observational studies
Encorafenib in combination with Cetuximab in BRAFV600E–Mutated Metastatic Colorectal Cancer: A European pooled analysis of observational studies
Encorafenib in combination with Cetuximab in BRAFV600E–Mutated Metastatic Colorectal Cancer: A European pooled analysis of observational studies.
How is the study conducted?
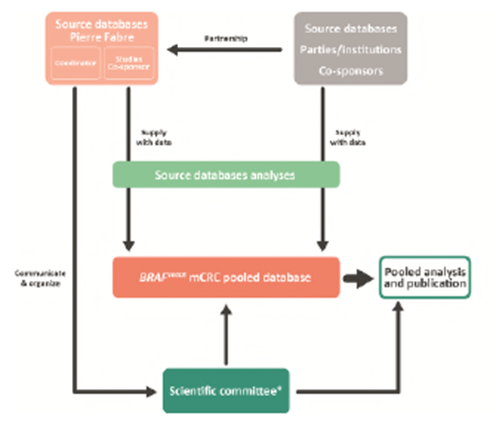
Who can take part in the trial ?
To be eligible to the study*, participant must fulfill several conditions including the following
- Patient aged over 18 years at encorafenib in combination with cetuximab initiation
- Patient having received encorafenib in combination with cetuximab for the treatment of BRAFV600E mutant mCRC.
Patients having received Encorafenib in combination with binimetinib treatment or enrolled in other trials are not part of the study.
*This is a pooled database of several databases. Enrolled patients are patients enrolled in the original studies
What are the objectives of the trial and how are they evaluated ?
The primary objective is to describe the clinical and demographic characteristics of BRAFV600E mutated mCRC patients treated with encorafenib in combination with cetuximab in a real-life setting across Europe.
Secondary objectives are :
- To describe the treatment patterns of BRAFV600E mutant mCRC patients receiving encorafenib in combination with cetuximab.
- To describe the therapeutic sequence,
- To describe the effectiveness in BRAFV600E mutant mCRC patients receiving encorafenib in combination with cetuximab.
- To describe the safety profile in BRAFV600E mutated mCRC patients receiving encorafenib in combination with cetuximab through the frequency of relevant AEs overall and by subgroup of patients (e.g. with/without baseline hepatic impairment).
- To identify prognostic factors specific to BRAFV600E mutant mCRC patients associated with OS and PFS.
- To identify potential clinical and biological factors associated with encorafenib in combination with cetuximab related toxicities.
- To describe BRAF mutation testing procedures, timing in relation to the line of treatment and turn-around time in real life settings.
